When fluid builds up around your lungs, it doesn’t just make breathing harder-it can signal something serious. This buildup, called pleural effusion, isn’t a disease on its own. It’s a symptom. And if you’re struggling to catch your breath, especially when lying down or moving, this might be why.
What Causes Pleural Effusion?
Not all fluid around the lungs is the same. Doctors split it into two types: transudative and exudative. The difference tells you what’s really going on inside your body. Transudative effusions happen when pressure or protein levels in your blood get out of balance. The most common cause? Congestive heart failure. About half of all pleural effusions come from this. When your heart can’t pump well, fluid backs up and leaks into the space around your lungs. Liver disease (cirrhosis) and kidney problems (nephrotic syndrome) can do the same thing. These are slow leaks, often linked to long-term conditions. Exudative effusions are more urgent. They happen when something is inflaming or damaging the pleura-the thin lining around your lungs. Pneumonia is the top culprit, causing 40-50% of these cases. Cancer is next, responsible for 25-30%. That’s why doctors take this seriously: if you have an exudative effusion, you might have an infection, a tumor, or a blood clot in the lung (pulmonary embolism). Tuberculosis still shows up in some areas, especially where it’s common. The key to figuring out which type you have? Light’s criteria. Developed in 1972, it’s still the gold standard. If your pleural fluid has a protein ratio over 0.5, LDH over 0.6 compared to blood, or LDH higher than two-thirds of the normal blood level-it’s exudative. These numbers don’t lie. And if your fluid pH is below 7.2, glucose under 60 mg/dL, or LDH over 1000 IU/L, you’re likely dealing with a complicated infection or cancer.How Thoracentesis Works
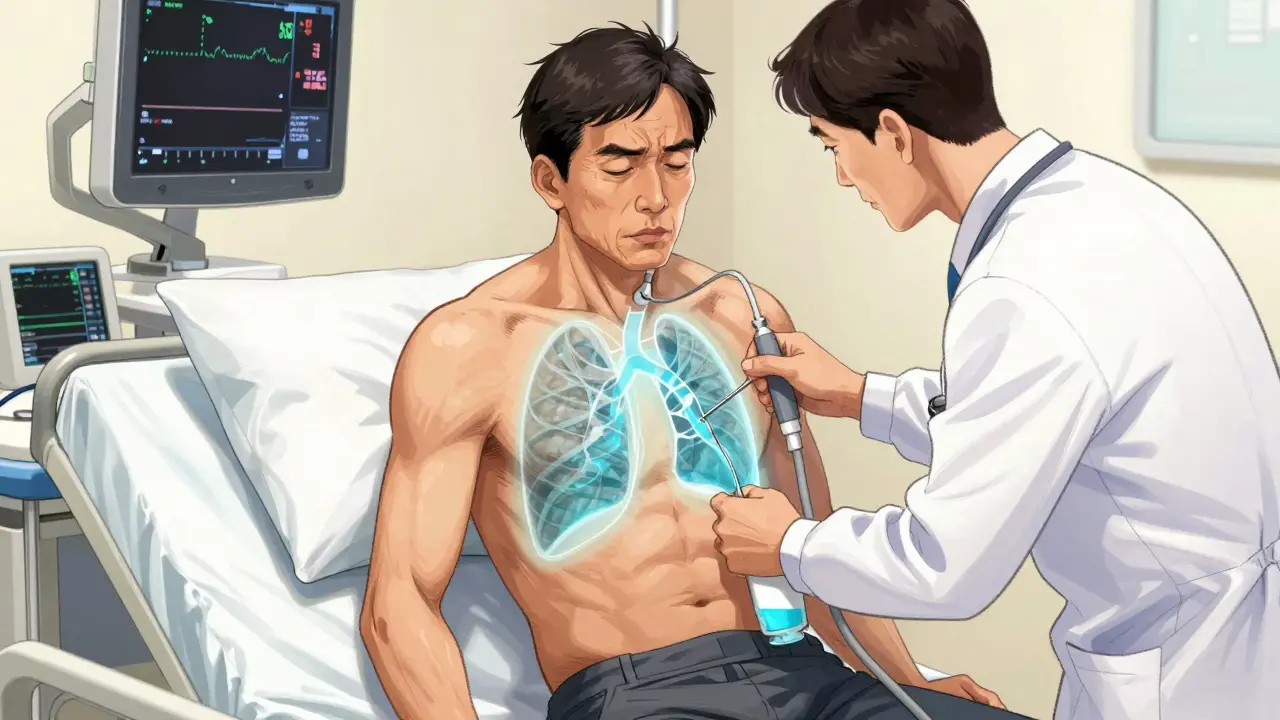
If fluid is building up and you’re short of breath, doctors don’t just wait. They drain it. That’s thoracentesis. It’s a simple procedure, but it’s not something you do without guidance. Ultrasound is now required. Before ultrasound, up to 19% of patients got a collapsed lung (pneumothorax) after the procedure. Now? That risk drops to under 5%. The ultrasound shows exactly where the fluid is, how much there is, and avoids hitting the lung or blood vessels. The needle or catheter goes in between the ribs, usually at the 5th to 7th space along the side of your chest. You’re sitting up, leaning forward. It takes less than 15 minutes. For diagnosis, they take 50-100 mL. For relief, they can safely remove up to 1500 mL in one go. But they don’t always drain it all. Too much too fast can cause re-expansion pulmonary edema-a rare but dangerous swelling of the lungs after they suddenly refill with air. That’s why some doctors now use pleural manometry: they measure pressure as they drain. If it stays under 15 cm H₂O, the risk of complications drops to less than 5%. The fluid gets tested. Protein, LDH, cell count, pH, glucose, and cytology (looking for cancer cells). Cytology finds cancer in about 60% of malignant cases. If glucose is low or pH is acidic, it points to an infection. High amylase? Could mean pancreatitis. Blood in the fluid? Might be a pulmonary embolism or trauma. Complications happen in 10-30% of cases without ultrasound. With it? Down to 4-6%. Pneumothorax is still the biggest risk, followed by bleeding and infection. But with proper technique, serious problems are rare.
Why You Can’t Just Drain and Walk Away
Draining fluid feels better. But if you don’t fix what caused it, the fluid comes back. Fast. For heart failure? The fix isn’t drainage-it’s medication. Diuretics like furosemide, ACE inhibitors, beta-blockers. When doctors use NT-pro-BNP levels to guide treatment, recurrence drops from 40% to under 15% in three months. Drain the fluid, but also treat the heart. For pneumonia? Antibiotics are the priority. If the fluid is thick, cloudy, or has a pH under 7.2 or glucose under 40 mg/dL, you need drainage. Left untreated, 30-40% of these turn into empyema-pus in the chest. That requires surgery. Now, the toughest one: cancer. Malignant pleural effusion has a 50% chance of coming back within 30 days after a single drainage. That’s why doctors don’t stop at thoracentesis. They go further.Preventing Recurrence: What Actually Works
The goal isn’t just to remove fluid. It’s to stop it from coming back. And the best method depends entirely on the cause. For cancer patients, two options dominate: pleurodesis and indwelling pleural catheters. Pleurodesis means sticking the lung to the chest wall. Talc slurry is the most common. It causes inflammation that fuses the layers together. Success? 70-90%. But it’s painful. About 60-80% of patients need strong pain meds afterward. Hospital stay? Usually 3-7 days. Indwelling pleural catheters (IPC) are changing the game. A small tube stays in place, usually for weeks. You drain it at home, a few times a week. No hospital stay. Less pain. Better quality of life. Success rate? 85-90% at six months. A 2021 study in the New England Journal of Medicine showed patients using IPCs went home in 2.1 days instead of 7.2. That’s huge. The American Thoracic Society now recommends IPCs as first-line for malignant effusions, especially if the lung is trapped or the patient has limited life expectancy. Talc is still good, but it’s no longer the only choice. For post-surgery effusions-common after heart bypass-most clear up on their own. But if more than 500 mL drains per day for three days straight, they keep the chest tube in. That prevents recurrence in 95% of cases. And here’s the catch: don’t drain small, silent effusions. A 2019 JAMA Internal Medicine study found 30% of thoracentesis procedures on small, asymptomatic effusions gave no benefit. No diagnosis. No relief. Just risk.
this post is fire🔥 i had pleural effusion last year and honestly thought i was dying. turned out it was just heart failure. drs drained me and i was like ‘wait, that’s it?’ now i take my diuretics like candy 💊 and breathe like a champ. thanks for the clarity!
The pathophysiological underpinnings of pleural effusion, as elucidated in this exposition, are both meticulously detailed and clinically pertinent. Transudative effusions, primarily attributable to hydrostatic pressure anomalies in congestive heart failure, necessitate a fundamentally different therapeutic paradigm than exudative effusions, which are invariably associated with inflammatory or neoplastic etiologies. The application of Light’s criteria remains, as of current evidence, the gold standard for differential diagnosis, and its continued utility underscores the enduring value of evidence-based clinical algorithms in modern medicine.
This is such an important topic! So many people think ‘shortness of breath’ is just getting older-but this breaks it down so clearly. I’ve seen patients panic over small effusions that didn’t need touching. The part about not draining asymptomatic fluid? That’s a game-changer. Keep sharing this kind of stuff. You’re helping people understand their bodies before they panic.
I find it profoundly concerning that modern medicine has become so procedure-driven. Drain this, inject that, stick a tube in-when will we return to treating the root? The notion that an indwelling catheter is preferable to pleurodesis for cancer patients speaks to a culture of convenience over cure. We are not repairing the body-we are managing its decay.
I appreciate how thoroughly this breaks down the clinical reasoning behind each intervention. The emphasis on ultrasound guidance is particularly well-placed-so many complications are preventable with proper imaging. I’ve seen cases where thoracentesis was performed blindly, and the outcomes were... not ideal. This is a textbook example of how precision improves safety.
I JUST got diagnosed with this last week and I’m sobbing reading this. I thought I was gonna die. I’m 32. No smoking. No heart issues. Then my doc says ‘it’s probably cancer’ and I lost it. But then I read the part about IPCs and I actually cried happy tears. I’m getting one next week. I’m gonna drain it at home like a boss. Thank you for this. Seriously.
I’m so glad someone mentioned the 2019 JAMA study. So many hospitals still drain tiny, asymptomatic effusions because ‘it’s easy’ or ‘we’re just checking.’ It’s not. It’s risky. And unnecessary. Please, if you’re a clinician reading this-don’t do it. If you’re a patient-ask if it’s truly needed. Don’t let fear drive your care.
You Americans always think you’ve invented medicine. Pleurodesis with talc? We’ve been doing this in Nigeria since the 1980s. And we didn’t have ultrasound. We had intuition. We had experience. We had men who knew anatomy because they didn’t rely on machines. The ‘gold standard’ you speak of? It’s just a fancy label for something our grandfathers did with a stethoscope and a needle. Modernity is not progress-it’s distraction.
Honestly? All this talk about ‘exudative’ and ‘transudative’ is just doctor jargon. Real talk: if you can’t breathe, get it out. Who cares what kind of fluid it is? You’re not a textbook. You’re a human. Stop overthinking. Just fix the problem. Also, why are we still using Light’s criteria from 1972? Shouldn’t AI be doing this by now?
I love how you included Dr. Light’s quote-‘bailing water from a sinking boat.’ That’s perfect. And yet, here we are, still bailing. We’ve got the tools to patch the hole-ultrasound, IPCs, biomarkers-but too many hospitals are stuck in 2005. The fact that 30% of thoracenteses are done on asymptomatic patients? That’s not medical practice. That’s fear-based billing. We need to stop treating symptoms and start treating people.
I’m a nurse. I’ve seen this happen so many times. A patient comes in, gasping, terrified. We do the thoracentesis. They feel amazing. They leave. Then they come back in two weeks. Same thing. And the family is like ‘why didn’t you fix it?’ But we didn’t fix it-we just made them feel better. And then they get mad because we didn’t ‘cure’ them. It’s heartbreaking. This post? This is the education we need to give patients BEFORE they get scared. Thank you.
I read this and thought: why is this so complicated? If the fluid’s back, the problem’s still there. Why not just leave the catheter in? Why the drama? Why the talc? Why the hospital stay? It’s like we’re scared of simplicity. Just put the tube in. Let people drain it. Let them live. Why make it a big event?
In India, we refer to pleural effusion as ‘paani phoolna’-water filling. Simple. Direct. The science is universal, but the language shouldn’t be. I’ve translated this entire post into Hindi for my uncle, who has CHF. He understood it. That’s what matters. Knowledge shouldn’t be locked behind English or jargon. Thank you for writing in a way that lets people actually learn.
The assertion that indwelling pleural catheters represent a superior modality for malignant effusions is not universally supported. While quality-of-life metrics are favorable, long-term survival outcomes remain equivocal. Moreover, the risk of catheter-related infection, particularly in immunocompromised hosts, is underreported in contemporary literature. The American Thoracic Society’s recommendation, while pragmatic, may reflect institutional bias toward cost containment rather than clinical superiority.